BASIC ELECTRICAL THEORY 1 next>
ATOM AND ITS FORCES
What is electricity? Electricity is defined as “the flow of
electrons through simple materials and devices” or “that force
which moves electrons.” Scientists think electricity is produced
by very tiny particles called electrons and protons. These
particles are too small to be seen, but exist as subatomic
particles in the atom. To understand how they exist, you must
first understand the structure of the atom.
The Atom
Elements are
the basic building blocks of all matter. The atom is the smallest
particle to which an element can be reduced while still keeping
the properties of that element. An atom consists of a positively
charged nucleus
surrounded by negatively charged electrons, so that
the atom as a whole is electrically neutral. The nucleus is
composed of two kinds of subatomic particles, protons and
neutrons, as shown in Figure 1. 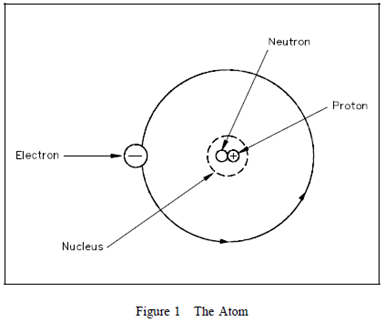 The proton
The proton
carries a single unit positive charge equal in magnitude to the
electron charge. The neutron is slighty heavier than the proton
and is electrically neutral, as the name implies. These two
particles exist in various combinations, depending upon the
element involved. The electron is the fundamental negative charge
(-) of electricity and revolves around the nucleus, or center, of
the atom in concentric orbits, or shells. The proton is the
fundamental positive charge (+) of electricity and is located in
the nucleus. The number of protons in the nucleus of any atom
specifies the atomic number of that atom or of that element. For
example, the carbon atom contains six protons in its nucleus;
therefore, the atomic number for carbon is six, as shown in
Figure 2. In its
In its
natural state, an atom of any element contains an equal number of
electrons and protons. The negative charge (-) of each electron
is equal in magnitude to the positive charge (+) of each proton;
therefore, the two opposite charges cancel, and the atom is said
to be electrically neutral, or in balance.
Electrostatic Force
One of the mysteries of the atom is that the electron and the
nucleus attract each other. This attraction is called
electrostatic force, the force that holds the electron in orbit.
This force may be illustrated with lines as shown in Figure
3.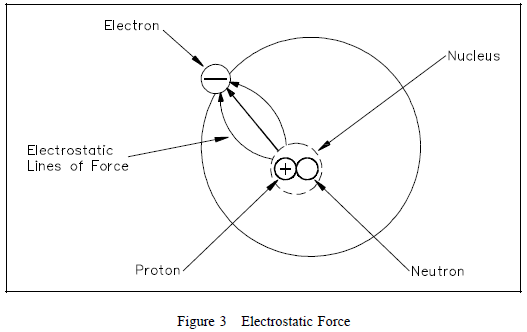 Without this
Without this
electrostatic force, the electron, which is traveling at high
speed, could not stay in its orbit. Bodies that attract each
other in this way are called charged bodies. As mentioned
previously, the electron has a negative charge, and the nucleus
(due to the proton) has a positive charge.
The First Law of Electrostatics
The negative charge of the electron is equal, but opposite to,
the positive charge of the proton. These charges are referred to
as electrostatic charges. In nature, unlike charges (like
electrons and protons) attract each other, and like charges repel
each other. These facts are known as the First Law
of Electrostatics
and are sometimes referred to as the law of electrical charges.
This law should be remembered because it is one of the vital
concepts in electricity. Some atoms can lose electrons and others
can gain electrons; thus, it is possible to transfer electrons
from one object to another. When this occurs, the equal
distribution of negative and positive charges no longer exists.
One object will contain an excess of electrons and become
negatively charged, and the other will become deficient in
electrons and become positively charged. These objects, which can
contain billions of atoms, will then follow the same law of
electrostatics as the electron and proton example shown above.
The electrons that can move around within an object are said to
be free electrons and will be discussed in more detail in a later
section. The greater the number of these free electrons an object
contains, the greater its negative electric charge. Thus, the
electric charge can be used as a measure of electrons.
Electrostatic Field
A special force is acting between the charged objects discussed
above. Forces of this type are the result of an electrostatic
field that exists around each charged particle or object. This
electrostatic field, and the force it creates, can be illustrated
with lines called “lines of force” as shown in Figure 4.
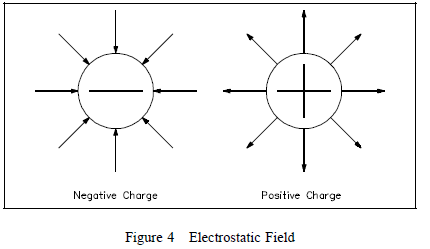 Charged
Charged
objects repel or attract each other because of the way these
fields act together. This force is present with every charged
object. When two objects of opposite charge are brought near one
another, the electrostatic field is concentrated in the area
between them, as shown in Figure 5. 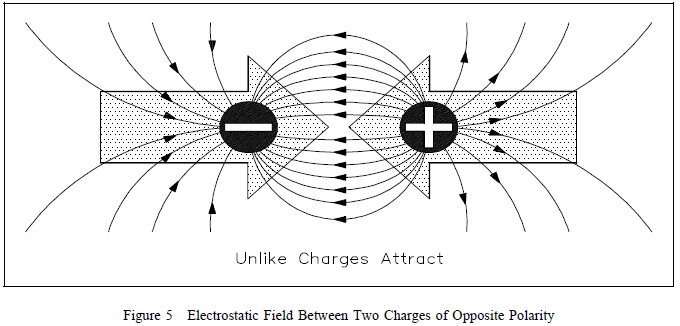 The direction
The direction
of the small arrows shows the direction of the force as it would
act upon an electron if it were released into the electric
field. When two objects of like charge are brought near one
another, the lines of force repel each other, as shown in Figure
6. 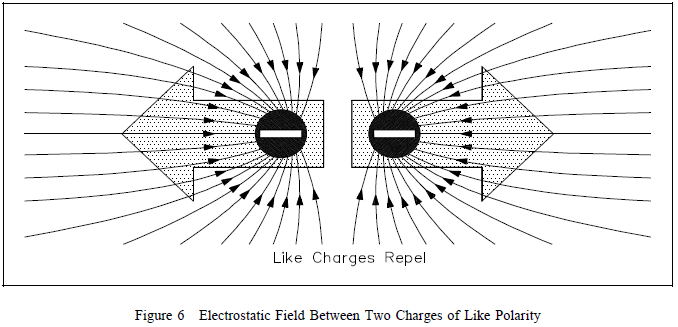 The strength
The strength
of the attraction or of the repulsion force depends upon two
factors: (1) the amount of charge on each object, and (2) the
distance between the objects. The greater the charge on the
objects, the greater the electrostatic field. The greater the
distance between the objects, the weaker the electrostatic field
between them, and vice versa. This leads us to the law of
electrostatic attraction, commonly referred to as Coulomb’s Law
of electrostatic charges, which states that the force of
electrostatic attraction, or repulsion, is directly proportional
to the product of the two charges and inversely proportional to
the square of the distance between them as shown in Equation 1-1.
Where F = force of electrostatic attraction or prepulsion
(Newtons) K = constant of proportionality (Coulomb 2/N-m2) q1 =
charge of first particle (Coulombs) q2 = charge of second
particle (Coulombs) d = distance between two particles (Meters)
If q1 and q2 are both either positively or negatively
charged, the force is repulsive. If q1 and q2 are opposite
polarity or charge, the force is attractive.
Potential Difference
Potential
difference is the term used to describe how large the
electrostatic force is between two charged objects. If a charged
body is placed between two objects with a potential difference,
the charged body will try to move in one direction, depending
upon the polarity of the object. If an electron is placed between
a negatively-charged body and a positively-charged body, the
action due to the potential difference is to push the electron
toward the positively-charged object. The electron, being
negatively charged, will be repelled from the
negatively-charged object and attracted by the
positively-charged object, as shown in Figure 7. 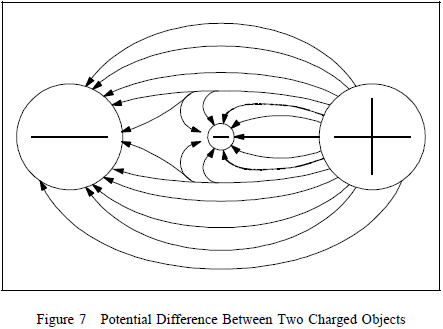 Due to the
Due to the
force of its electrostatic field, these electrical charges have
the ability to do work by moving another charged particle by
attraction and/or repulsion. This ability to do work is called
“potential”; therefore, if one charge is different from another,
there is a potential difference between them. The sum of the
potential differences of all charged particles in the
electrostatic field is referred to as electromotive force (EMF).
The basic unit of measure of potential difference is the “volt.”
The symbol for potential difference is “V,” indicating the
ability to do the work of forcing electrons to move. Because the
volt unit is used, potential difference is also called “voltage.”
The unit volt will be covered in greater detail in the next
chapter.
Free Electrons
Electrons are in rapid motion around the nucleus. While the
electrostatic force is trying to pull the nucleus and the
electron together, the electron is in motion and trying to pull
away. These two effects balance, keeping the electron in orbit.
The electrons in an atom exist in different energy levels. The
energy level of an electron is proportional to its distance from
the nucleus. Higher energy level electrons exist in orbits, or
shells, that are farther away from the nucleus. These shells nest
inside one another and surround the nucleus. The nucleus is the
center of all the shells. The shells are lettered beginning with
the shell nearest the nucleus: K, L, M, N, O, P, and Q. Each
shell has a maximum number of electrons it can hold. For example,
the K shell will hold a maximum of two electrons and the L shell
will hold a maximum of eight electrons. As shown in Figure 8,
each shell has a specific number of electrons that it will hold
for a particular atom.
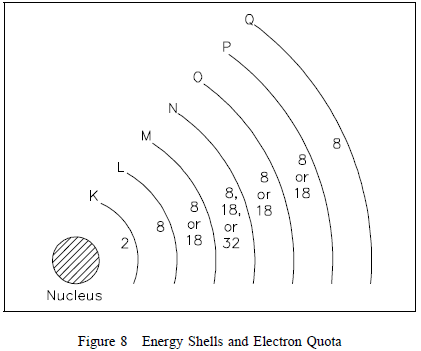
There are two simple rules concerning electron shells that make
it possible to predict the electron distribution of any element:
1.The maximum number of electrons that can fit in the outermost
shell of any atom is 8. 2. The maximum number of electrons that
can fit in the next-to-outermost shell of any atom is 18. An
important point to remember is that when the outer shell of an
atom contains eight electrons, the atom becomes very stable, or
very resistant to changes in its structure. This also means that
atoms with one or two electrons in their outer shell can lose
electrons much more easily than atoms with full outer shells. The
electrons in the outermost shell are called valence
electrons. When external energy, such as heat, light, or
electrical energy, is applied to certain materials, the
electrons gain energy, become excited, and may move to a higher
energy level. If enough energy is applied to the atom, some of
the valence electrons will leave the atom. These electrons are
called free
electrons. It is the movement of free electrons that provides
electric current in a metal conductor. An atom that has lost or
gained one or more electrons is said to be ionized or to have
an ion change. If the atom loses one or more electrons, it
becomes positively charged and is referred to as a positive ion.
If an atom gains one or more electrons, it becomes negatively
charged and is referred to as a negative ion.
BASIC ELECTRICAL THEORY 1 next>
